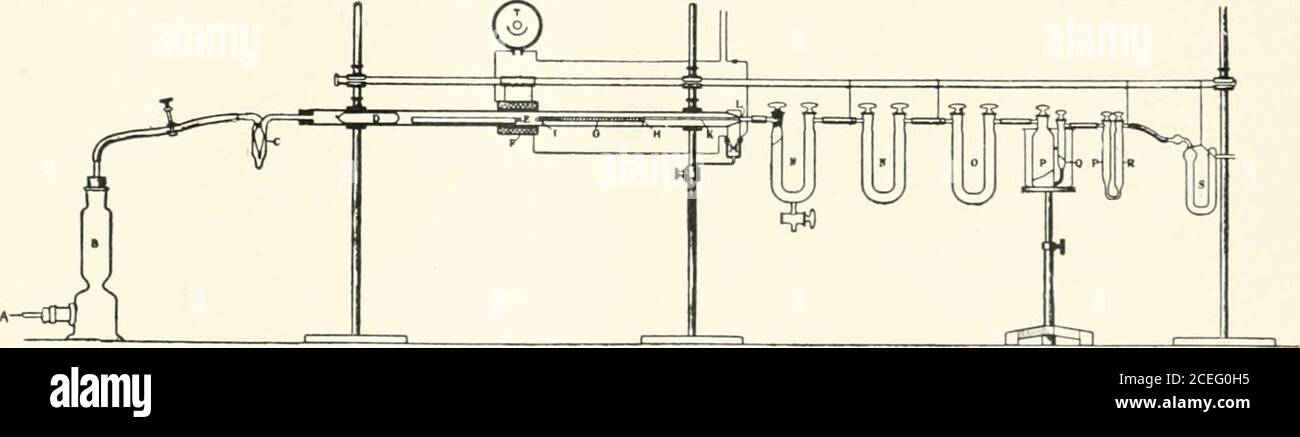
India rubber world. ning alundum,and after the acetone and the ethyl acetate have been expelledby warming the boat for several hours in the drying oventhe nitrosite is burned in a current

Diluente Nitro Antinebbia Lt.25 UN1993, Liquido infiammabile N.A.S. (Acetone; N-eptano),3, II (D/E) Pericoloso per l'ambiente – Gst Italia

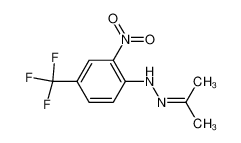
Figure 2 | Synthesis and Conformational Assignment of N-(E)-Stilbenyloxymethylenecarbonyl-Substituted Hydrazones of Acetone and o-(m- and p-) Chloro- (nitro-) benzaldehydes by Means of and NMR Spectroscopy

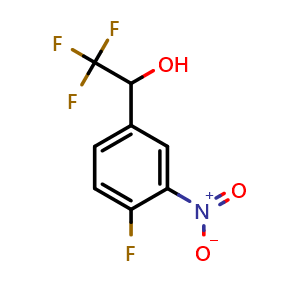
Synthesis of 4-Hydroxy-4-(4-nitrophenyl)butan-2-one using p-Nitro Benzaldehyde and Acetone in Aqueous Micellar Media using L-Proline | Semantic Scholar