Form FDA 3455 - Disclosure: Financial Interest and Arrangements of Clinical Investigators Free Download

Financial Disclosure Form Clinical Trials Template - Fill Online, Printable, Fillable, Blank | pdfFiller
SECTION: Research and Compliance NUMBER: CLN-21.00 CHAPTER: Clinical Research Standard Operating Procedures 7/28/2008 REV. A: 5/

FDA: Financial Disclosure by Clinical Investigators (Draft Guidance) | PDF | Clinical Trial | Food And Drug Administration
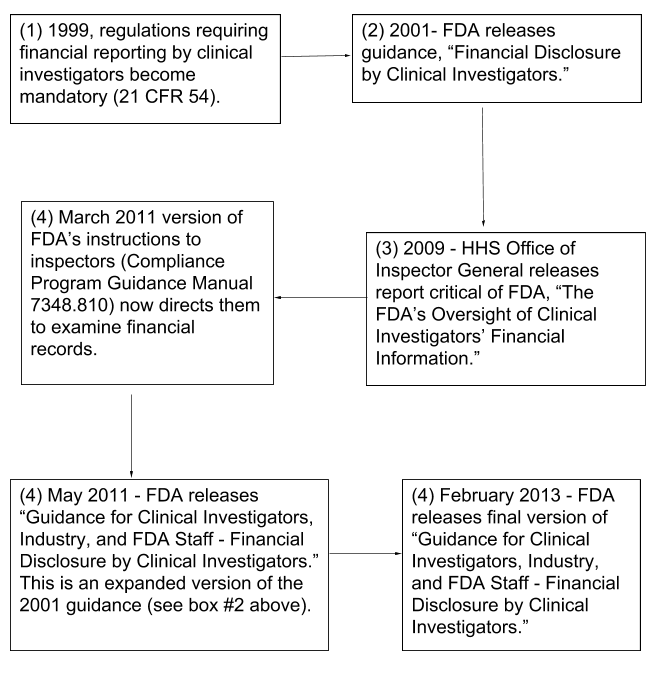
Process for collection of financial disclosure by clinical investigators per 21 CFR 54.4 Background: U.S. regulations, 21 CFR 32