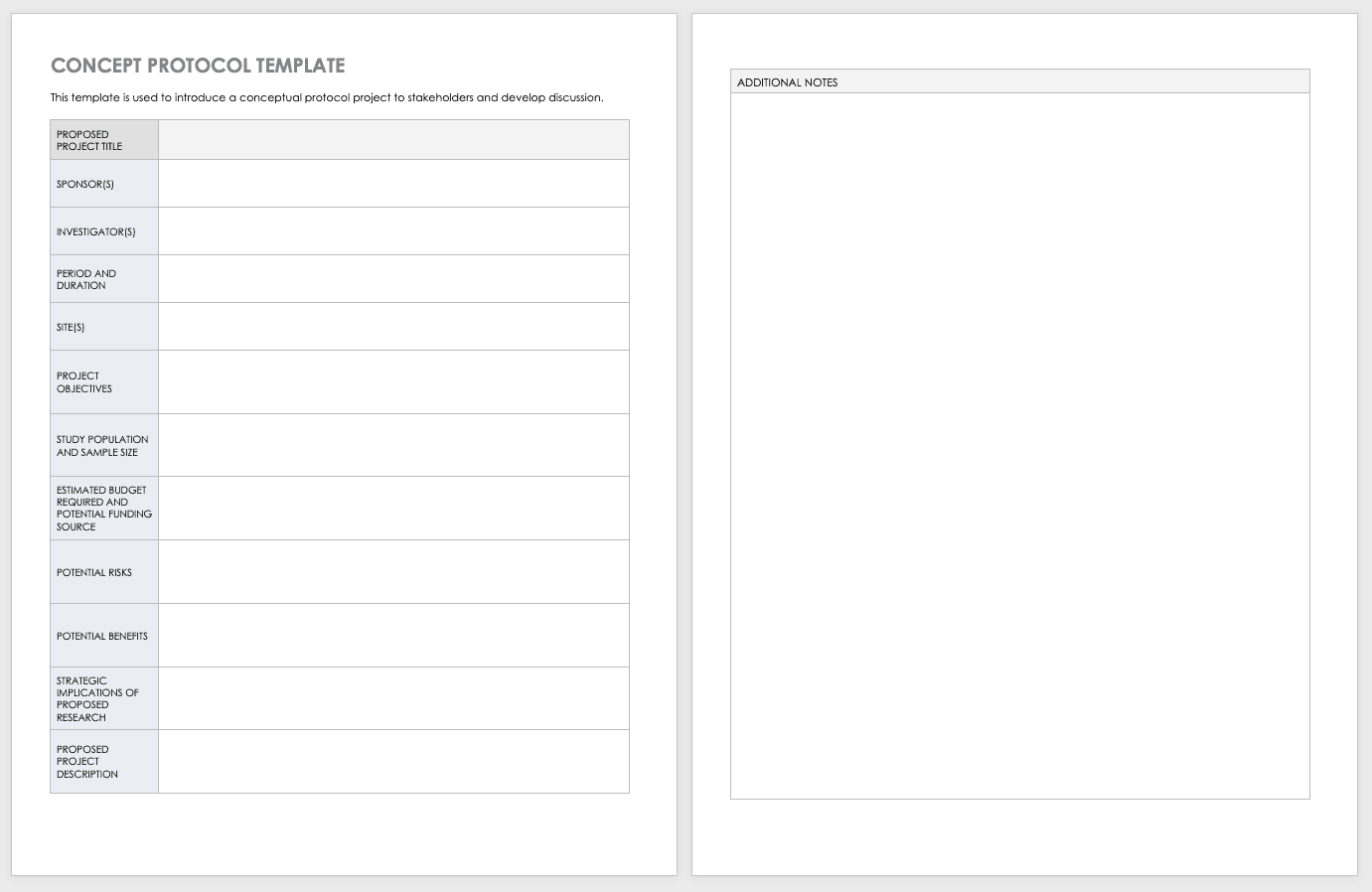
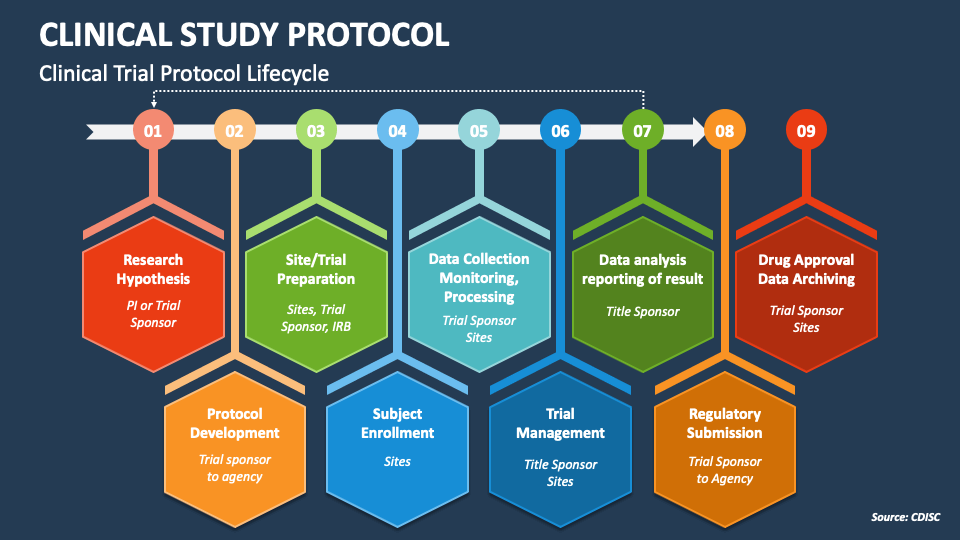
NIH-FDA Phase 2 and 3 IND/IDE Clinical Trial Protocol Template PREFACE Remove this Preface before finalizing and distributing
TEMPLATE FOR THE CLINICAL ASSESSMENT OF CLINICAL TRIAL APPLICATIONS African Vaccine Regulatory Forum (AVAREF) CLINICAL ASSESSMEN

Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension - The Lancet Digital Health

![PDF] Designing a Case Study Protocol for Application in IS Research | Semantic Scholar PDF] Designing a Case Study Protocol for Application in IS Research | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/bf2a1603faa76132c097653066392f653c106b27/10-Table2-1.png)